 
This cookie is set by GDPR Cookie Consent plugin. The cookie is set by GDPR cookie consent to record the user consent for the cookies in the category "Functional". The cookie is used to store the user consent for the cookies in the category "Analytics". These cookies ensure basic functionalities and security features of the website, anonymously. Necessary cookies are absolutely essential for the website to function properly. The basic formula for the enthalpy of formation is the sum of the enthalpy of the reactants subtracted from… The enthalpy of formation is defined as the energy, in the form of heat, given off or absorbed during a chemical reaction under the most stable conditions for the reaction to occur from pure elements. What is the definition of enthalpy of formation? The standard enthalpy of reaction, ΔH⊖ rxn Δ H r x n ⊖, can be calculated by summing the standard enthalpies of formation of the reactants and subtracting the value from the sum of the standard enthalpies of formation of the products. What is formula for standard enthalpy of formation? Heats of formation can be determined in the following ways: through direct calorimetric measurement by using the equations describing the constant-pressure and constant-volume temperature dependence of the equilibrium constant of the formation reaction by calculation, on the basis of Hess’s law, from the heat of a reaction in which the given How do you calculate the heat of formation? A given reaction is considered as the decomposition of all reactants into elements in their standard states, followed by the formation of all products. The standard enthalpy change of any reaction can be calculated from the standard enthalpies of formation of reactants and products using Hess’s law. How do you calculate standard enthalpy of formation? At standard conditions hydrogen is a gas of diatomic molecules having the formula H2…. Hydrogen is the chemical element with the symbol H and atomic number 1. The formation of water from H2 (g) and O2 (g) is an exothermic reaction because.ĭoes hydrogen have enthalpy of formation? A pure element in its standard state has a standard enthalpy of formation of zero. 
The standard enthalpy of formation of a substance is the enthalpy change that occurs when 1 mole of the substance is formed from its constituent elements in their standard states. 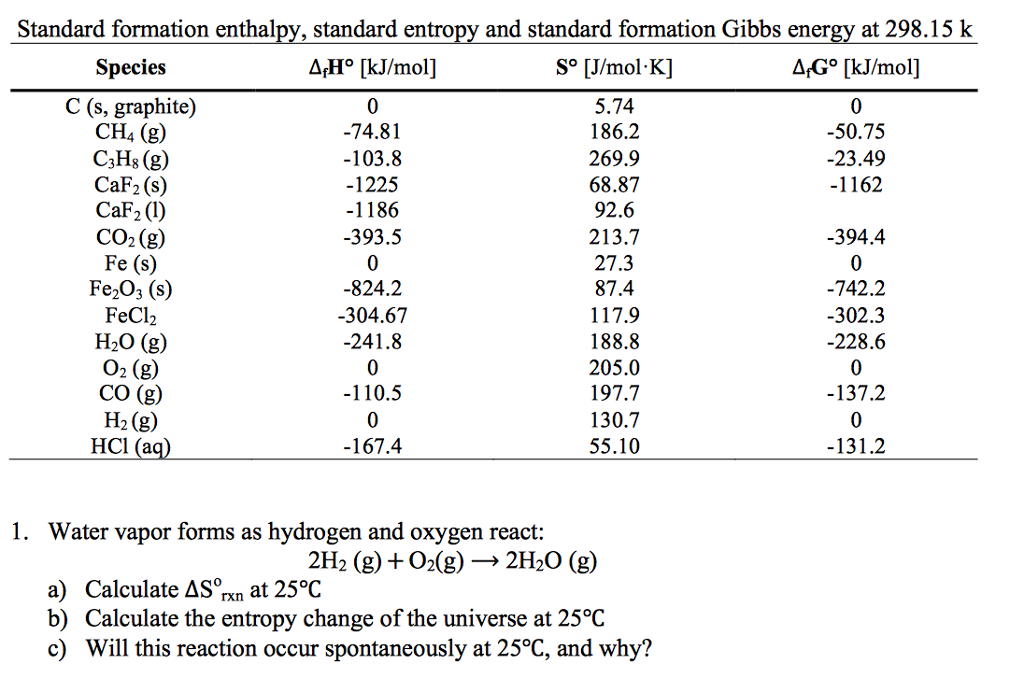
What is the standard enthalpy of formation of an element? and the standard enthalpy of formation values: ΔH fo = 433 KJ/mol. This equation essentially states that the standard enthalpy change of formation is equal to the sum of the standard enthalpies of formation of the products minus the sum of the standard enthalpies of formation of the reactants. How do you find the standard enthalpy of formation? Molar Enthalpy of Formation of Various Substances SubstanceĮnthalpy of hydrogenation, defined as the enthalpy change observed in a constituent of a thermodynamic system when one mole of an unsaturated compound reacts completely with an excess of hydrogen to form a saturated compound. Therefore, O 2(g), H 2(g), and graphite have ΔHof values of zero. For example, although oxygen can exist as ozone (O 3), atomic oxygen (O), and molecular oxygen (O 2), O 2 is the most stable form at 1 atm pressure and 25☌. The standard enthalpy of formation of any element in its standard state is zero by definition. Table 20.2.1: Standard Entropy Values at 25oCĭoes hydrogen have an enthalpy of formation?įor H2 enthalpy of formation is zero, because it already is the most elementary form.ĭoes H2 have a standard enthalpy of formation of zero? The symbol for entropy is S and the standard entropy of a substance is given by the symbol So, indicating that the standard entropy is determined under standard conditions….Standard Entropy. Now, the enthalpy change for a formation reaction is called enthalpy of formation. The enthalpy of formation for an element in its elemental state will always be 0 because it takes no energy to form a naturally-occurring compound. Why is enthalpy of formation of hydrogen 0? 8 How do you calculate the heat of formation?.7 How do you calculate standard enthalpy of formation?.1 Why is enthalpy of formation of hydrogen 0?. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
